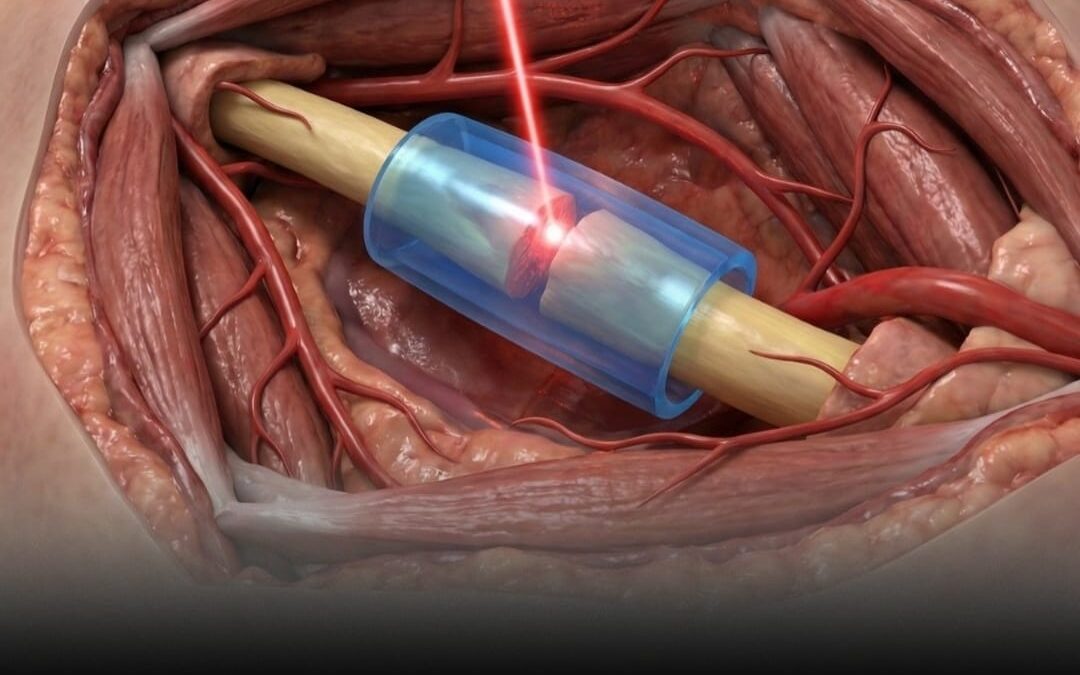
The FDA has approved a revolutionary light-activated polymer system that repairs severed nerves without the need for traditional surgical stitches.
The US Food and Drug Administration has officially cleared a revolutionary nerve-repair system that replaces traditional surgical stitches with light-activated polymers. Developed by medical technology firm Tissium based on research from MIT, this innovation utilizes a biocompatible polymer to create a flexible, 3D-printed cuff around severed nerves. Instead of the painstaking process of manual suturing under a microscope, surgeons can now apply a liquid prepolymer that cures instantly with light, creating a secure seal that allows delicate nerve tissues to knit back together naturally.
This milestone marks the first FDA approval for Tissium, culminating over a decade of research into poly(glycerol sebacate acrylate) materials. The implant is designed to be fully bioresorbable, meaning it safely disappears once the nerve has healed, reducing the risk of inflammation or complications from permanent foreign materials. Beyond nerve reconstruction, the company is already scaling its manufacturing capabilities to adapt this technology for cardiovascular sealing and gastrointestinal hernia repairs, aiming to transform complex microsurgeries into streamlined, highly effective clinical solutions.
source: Howes, L. (2025). FDA approves light-activated polymer for nerve repair without stitches. Chemical & Engineering News.